Abstract
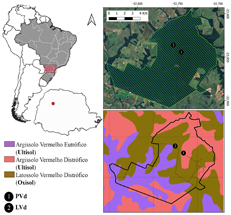
The Atlantic Forest is one of the most biodiverse biomes in the world. A significant protected area in Paraná State (Brazil) represents an ecotone between two subdivisions: the Upper Paraná Atlantic Forest and the Araucaria Moist Forest. This protected area experiences a loss of approximately half of its canopy tree leaves between June and August, leading to the formation of a thick layer of leaf litter. This environment fosters a high abundance and diversity of Staphylinidae beetles, the beetle family with the highest species richness, particularly in the Neotropics. This study investigated whether soil characteristics influence Staphylinidae composition and diversity. Among the two soil types analyzed, Ultisol exhibited the highest overall richness (63 morphospecies) and diversity, whereas Oxisol (38 morphospecies) hosted a compositionally distinct community with greater internal variability, a pattern consistent across years. A generalized linear model confirmed soil type as the main predictor of richness, while NMDS ordination showed distinct community clustering by soil type, corroborated by PERMANOVA. These results infer the importance of Ultisol within the reserve, given that it is subject to greater disturbance through direct contact with crops and highways. Demonstrating the importance of soil properties in biodiversity monitoring enhances the understanding of habitat heterogeneity and reinforces the relevance of soil health.
References
Arenhardt T. C. P., Vitorino M. D. & Martins S. V. 2025. Community structure of Coleoptera families and Staphylinidae species as potential bioindicators in Atlantic rain forest. Floresta e Ambiente 31(4): e20240029. https://doi.org/10.1590/2179-8087-FLORAM-2024-0029
Asenjo A., Klimaszewski J., Chandler D. S., Fierros-López H. E. & Vieira J. S. 2019. Staphylinidae (Insecta: Coleoptera) in Latin America: synopsis, annotated catalog, diversity and distribution. Zootaxa 4621(1): 1–406. https://doi.org/10.11646/zootaxa.4621.1.1
Ashford O. S., Foster W. A., Turner B. L., Sayer E. J., Sutcliffe L. & Tanner E. V. J. 2013. Litter manipulation and the soil arthropod community in a lowland tropical rainforest. Soil Biology and Biochemistry 62: 5–12. https://doi.org/10.1016/j.soilbio.2013.03.001
Beng K. C., Corlett R. T. & Tomlinson K. W. 2018. Seasonal changes in the diversity and composition of the litter fauna in native forests and rubber plantations. Scientific Reports 8: 10232. https://doi.org/10.1038/s41598-018-28603-7
Besuchet C., Burckhardt D. H. & Löbl I. 1987. The Winkler/Moczarski eclector as efficient extractor for fungus and litter Coleoptera. The Coleopterists Bulletin 41: 392-394.
Bitetti M. S., Placci G., & Dietz L. A. 2003. A biodiversity vision for the Upper Paraná Atlantic Forest ecoregion: designing a biodiversity conservation landscape and setting priorities for conservation action. WWF, Washington, 148 pp.
Caron E., Monné M. L., Ferreira V. S., Costa C., Cupello M., Aloquio S. & Tomaszewska W. 2024. Coleoptera of Brazil: what we knew then and what we know now. Insights from the Catálogo Taxonômico da Fauna do Brasil. Zoologia (Curitiba) 41(1): e23072 https://doi.org/10.1590/S1984-4689.v41.e23072
Castella P. R. & Britez R. M. (Orgs.). 2004. A Floresta com Araucária no Paraná: Conservação e diagnóstico dos remanescentes florestais. Brasília: Ministério do Meio Ambiente. 236 pp.
Caviglione J. H., Kiihl L. R. B., Caramori P. H., Oliveira D., Galdino J., Borrozino E. & Pugsley L. 2000. Cartas climáticas do Estado do Paraná. Iapar, Londrina, 1pp.
Chao A., Gotelli N. J., Hsieh T. C., Sander E. L., Ma K. H., Colwell R. K. & Ellison A. M. 2014. Rarefaction and extrapolation with Hill numbers: a framework for sampling and estimation in species diversity studies. Ecological Monographs 84: 45–67. https://doi.org/10.1890/13-0133.1
Crowder D. W. & Snyder W. E. 2010. Eating their way to the top? Mechanisms underlying the success of invasive insect generalist predators. Biological Invasions 12(9): 2857–2876. https://doi.org/10.1007/s10530-010-9733-8
Del Toro I., Ribbons R. R. & Pelini S. L. 2012. The little things that run the world revisited: a review of ant-mediated ecosystem services and disservices (Hymenoptera: Formicidae). Myrmecological News 17(0): 133–146.
Dias H. T., Figueira M. D., Silveira V., Fontes M. A. L., Oliveira Filho A. T. & Scolforo J. R. S. 2002. Variação temporal de nutrientes na serapilheira de um fragmento de floresta estacional semidecidual montana em Lavras. Cerne 8(2): 1–17.
Fujii K., Shibata M., Kitajima K., Ichie, T., Kitayama K. & Turner B. L. 2018. Plant–soil interactions maintain biodiversity and functions of tropical forest ecosystems. Ecological Research 33(1): 149–160. https://doi.org/10.1007/s11284-017-1511-y
Hopp F. W., Caron E., Ottermanns R. & Ross-Nickoll M. 2011. Evaluating leaf litter beetle data sampled by Winkler extraction from Atlantic Forest sites in southern Brazil. Revista Brasileira de Entomologia 55: 253–266. https://doi.org/10.1590/S0085-56262011000200017
Hopp F. W., Ottermanns R., Caron E., Meyer S. & Ross-Nickoll M. 2010. Recovery of litter inhabiting beetle assemblages during forest regeneration in the Atlantic forest of Southern Brazil. Insect Conservation and Diversity 3: 103–113. https://doi.org/10.1111/j.1752-4598.2010.00078.x
Instituto Brasileiro de Geografia e Estatística (IBGE). 2012. Manuais técnicos em geociências. IBGE, Rio de Janeiro, 978-85-240-4272-0, ISSN 0103-9598.
Irmler U., Klimaszewski J. & Betz O. 2018. Introduction to the Biology of Rove Beetles, pp. 1-X. In: Biology of Rove Beetles (Staphylinidae). Vol, 1 (O. Betz, U. Irmler, J. Klimaszewski, editors). Springer, Cham. https://doi.org/10.1007/978-3-319-70257-5_1
Kardol P., Cregger M. A., Campany C. E. & Classen A. T. 2010. Soil ecosystem functioning under climate change: plant species and community effects. Ecology 91: 767–781. https://doi.org/10.1890/09-0135.1
Lassau S. A., Hochuli D. F., Cassis G., & Reid C. A. 2005. Effects of habitat complexity on forest beetle diversity: do functional groups respond consistently. Diversity and distributions 11(1): 73–82. https://doi.org/10.1111/j.1366-9516.2005.00124.x
Laurance W. F. 2008. Conserving the hottest of the hotspots. Biological Conservation 142: 1137. https://doi:10.1016/j.biocon.2008.10.011
Magurran A. E. 2003. Measuring biological diversity. John Wiley & Sons, Oxford, 256 pp.
Méndez-Rojas D. M., Cultid-Medina C. & Escobar F. 2021. Influence of land use change on rove beetle diversity: A systematic review and global meta-analysis of a mega-diverse insect group. Ecological Indicators 122: 107239. https://doi.org/10.1016/j.ecolind.2020.107239
Montanari R. & Mário N. E. 2024. Classificação de latossolos e argissolos em diferentes pedoformas. Observatorio de la Economía Latinoamericana 22(12): e8150-e8150. https://doi.org/10.55905/oelv22n12-086
Navarrete-Heredia J. L. & Newton A. F. 2002. Guía ilustrada para los géneros de Staphylinidae (Coleoptera) de México. Biota Colomb 4: 271–273.
Oksanen J., Simpson G. L., Blanchet F. G., Kindt R., Legendre P., Minchin P. R., O’Hara R. B., Solymos P., Stevens M. H. H., Szoecs E., Wagner H., Barbour M., Bedward M., Bolker B., Borcard D., Borman T., Carvalho G., Chirico M., De Caceres M., Durand S., Evangelista H. B. A., FitzJohn R., Friendly M., Furneaux B., Hannigan G., Hill M. O., Lahti L., Martino C., McGlinn D., Ouellette M.-H., Ribeiro Cunha E., Smith T., Stier A., ter Braak C. J. F. & Weedon J. 2025. vegan: Community Ecology Package. Version 2.7-2. R Foundation for Statistical Computing, Vienna. https://doi.org/10.32614/CRAN.package.vegan
Ottermanns R., Hopp P. W., Guschal M., Santos G. P., Meyer S. & Roß-Nickoll M. 2011. Causal relationship between leaf litter beetle communities and regeneration patterns of vegetation in the Atlantic rainforest of Southern Brazil (Mata Atlântica). Ecological Complexity 8: 299–309. https://doi.org/10.1016/j.ecocom.2011.06.001
Rigal F., Cardoso P., Lobo J. M., Triantis K. A., Whittaker R. J., Amorim I. R. & Borges P. A. 2018. Functional traits of indigenous and exotic ground‐dwelling arthropods show contrasting responses to land‐use change in an oceanic island, Terceira, Azores. Diversity and Distributions 24(1): 36–47. https://doi.org/10.1111/ddi.12655
Roderjan C. V., Galvão F., Kuniyoshi Y. S. & Hatschbach G. G. 2002. As regiões fitogeográficas do Estado do Paraná. Revista Ciência e Ambiente 24: 75–92.
Rusch A., Birkhofer K., Bommarco R., Smith H. G. & Ekbom B. 2014. Management intensity at field and landscape levels affects the structure of generalist predator communities. Oecologia 175(3): 971–983. https://doi.org/10.1007/s00442-014-2949-z
Santos H. G., Jacomine P. K. T., Anjos L. H. C., Oliveira V. A., Lumbreras J. F., Coelho M. R., Almeida J. A., Araújo Filho J. C., Oliveira J. B. & Cunha T. J. F. 2025. Sistema Brasileiro de Classificação de Solos. Embrapa, 6 Edition. Brasília. ISBN 978-65-5467-104-0
Tews J., Brose U., Grimm V., Tielbörger K., Wichmann M. C., Schwager M. & Jeltsch F. 2004. Animal species diversity driven by habitat heterogeneity/diversity: the importance of keystone structures. Journal of biogeography 31(1): 79–92. https://doi.org/10.1046/j.0305-0270.2003.00994.x
Vasconcelos H. L. & Bruna E. M. 2012. Arthropod responses to the experimental isolation of Amazonian forest fragments. Zoologia (Curitiba) 29: 515–530. https://doi.org/10.1590/S1984-46702012000600003
Vijith H. & Dodge-Wan D. 2020. Spatial and temporal characteristics of rainfall over a forested river basin in NW Borneo. Meteorology and Atmospheric Physics 132: 683–702. https://doi.org/10.1007/s00703-019-00714-4